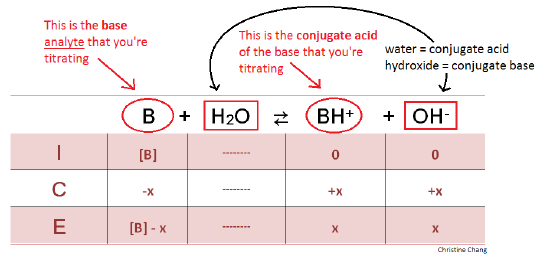
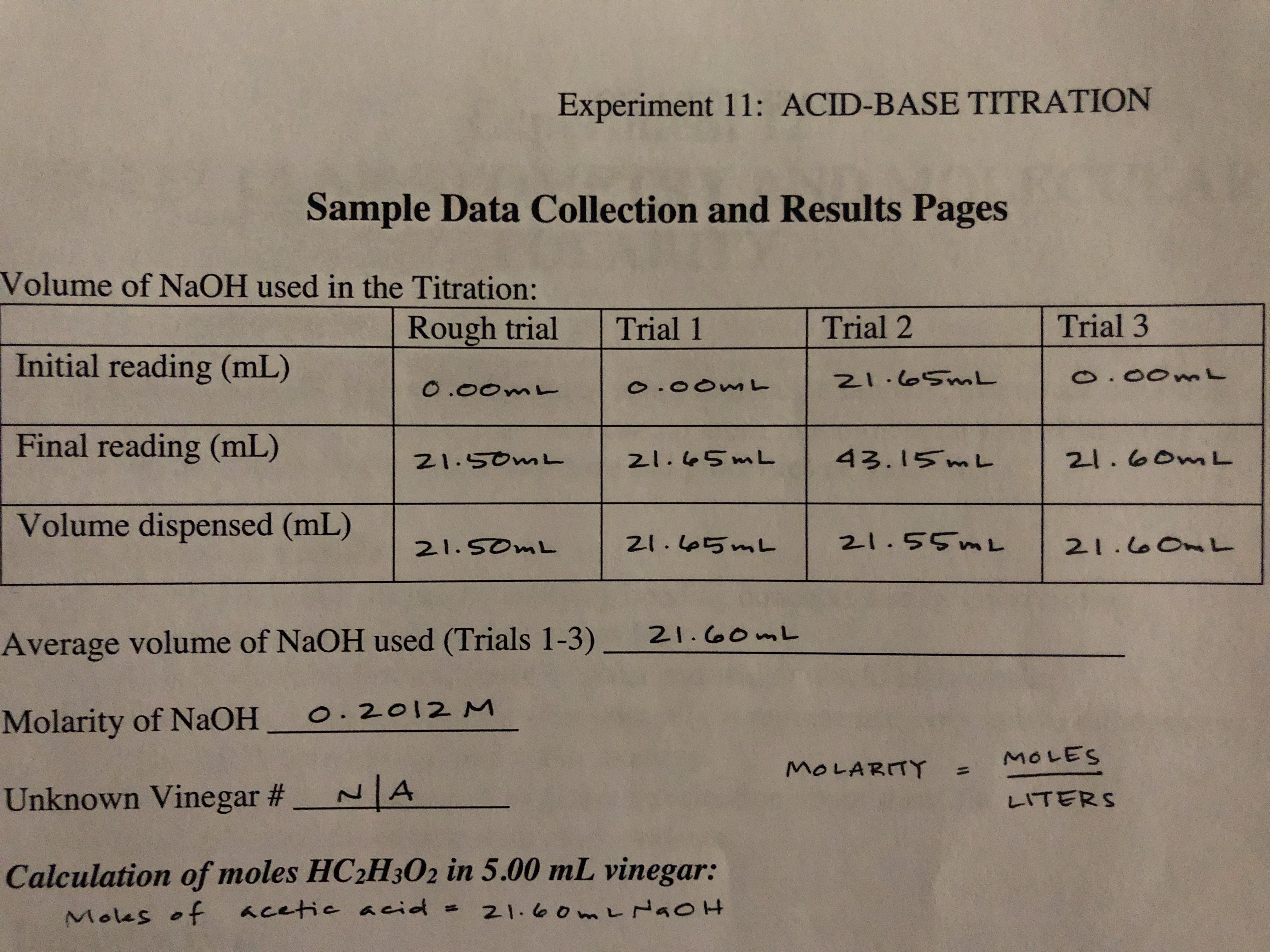
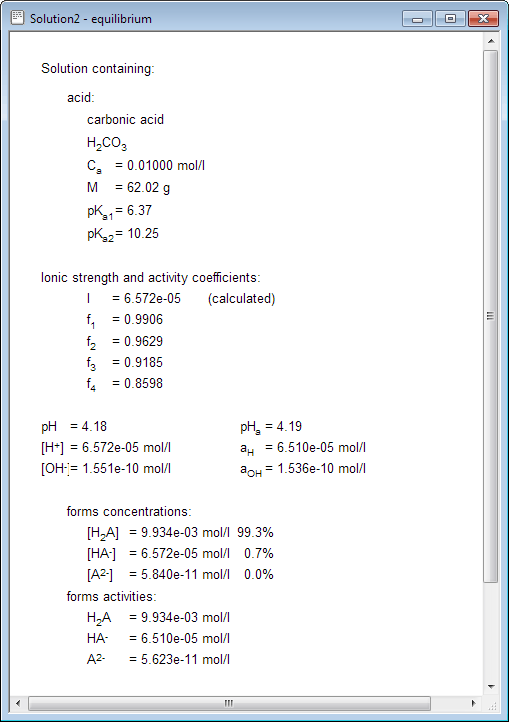
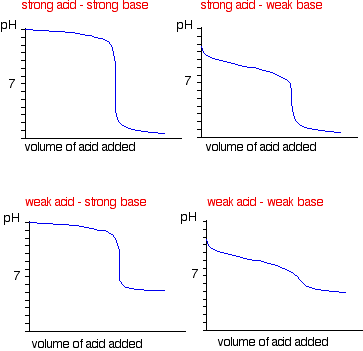
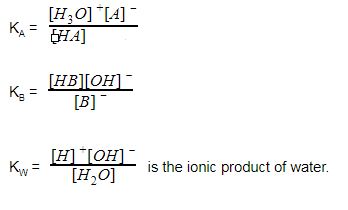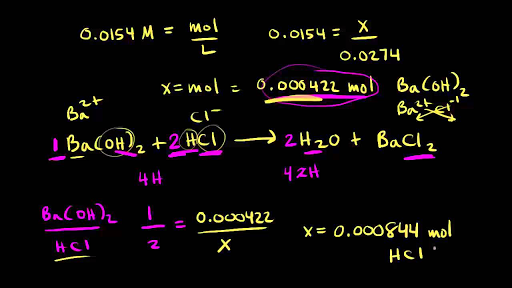equilibrium - How to calculate the dissociation constant of a weak acid from the titration with a strong base? - Chemistry Stack Exchange

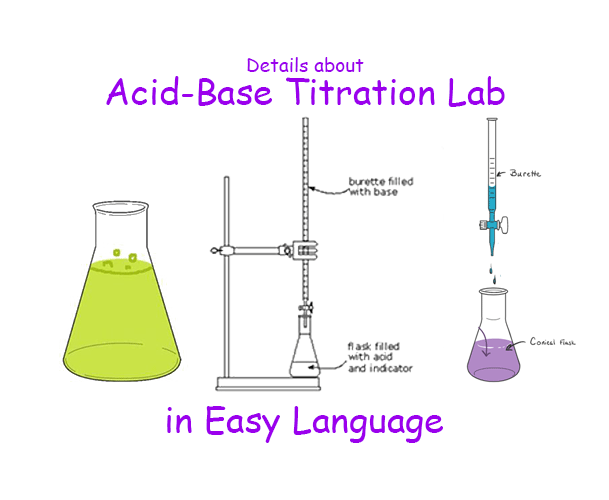
Acid-alkali volumetric titrations calculating concentrations from experimental results, apparatus, indicator, conical flask, pipette, burette gcse chemistry Calculations antacid indigestion tablet igcse KS4 science A level GCE AS A2 O Level practice ...

11.8 Acid–Base Titration The titration of an acid. A known volume of an acid is placed in a flask with an indicator and titrated with a measured volume. - ppt download

How to Calculate Analyte Concentration Using the Equivalence Point in an Acid-base Titration | Chemistry | Study.com