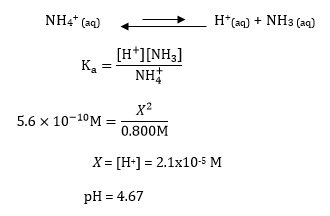
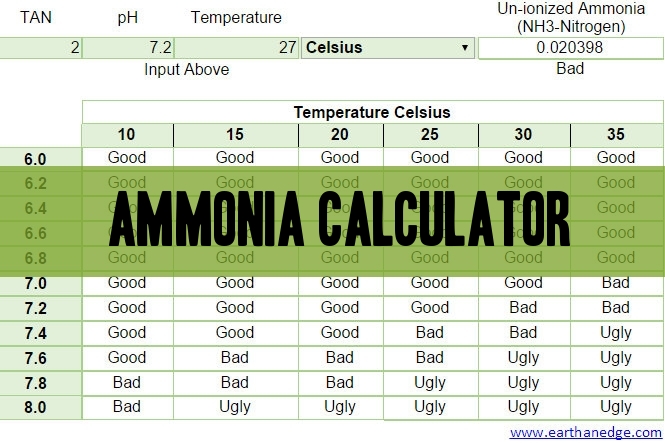
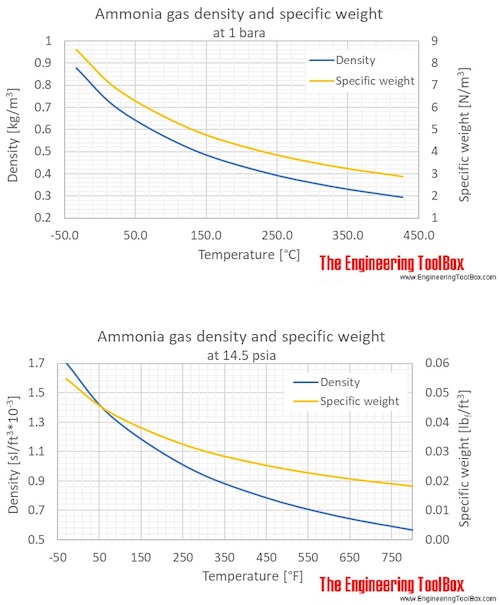
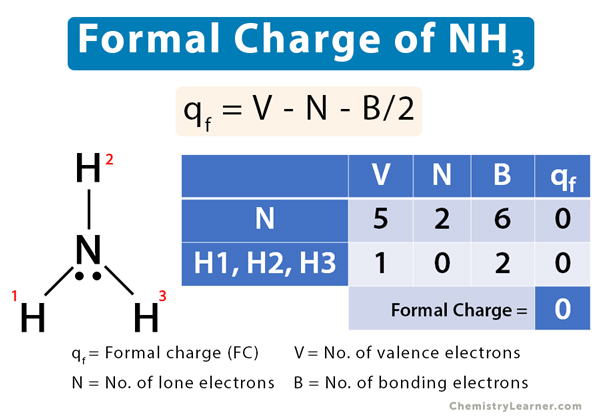
Step-by-step guide to calculating unionized (toxic) ammonia. UIA of... | Download Scientific Diagram

Calculate the equilibrium constant (Kc) for the formation of NH3 in the following reaction: N2 (g) + 3H2 (g) 2NH3 (g) At equilibrium, the concentration of NH3, H2 and N2 are 1.2 ×

Question Video: Calculating the Equilibrium Constant for Concentration Given the Initial Amount of Each Reactant | Nagwa
A gas jar contains 1.7 g of ammonia gas. Calculate the following: (i) Molar mass of ammonia. (ii) How many moles of ammonia are present in the gas jar ? (iii) How